 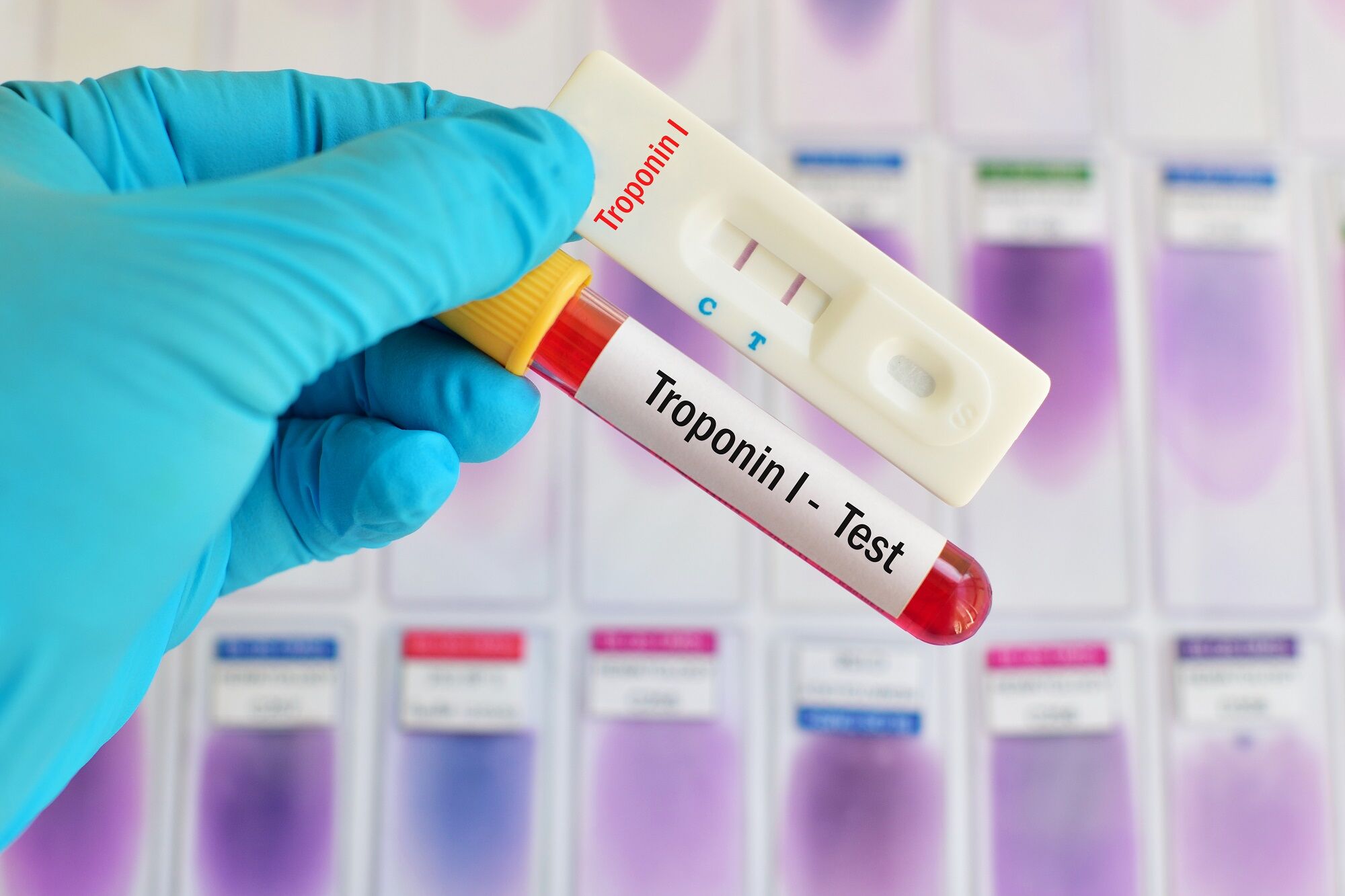
For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics. the performance of the i-STAT POC troponin was tested using a cut point of 0.080ng/mL, which corre- sponds to the package insert 99th percentile URL for. Check with your local representative for availability in specific markets. Not all products are available in all regions. GDPR Statement | Declaration for California Compliance Law. Using i-STAT troponin Normal 0.04 microgram/L Elevated 0.04 microgram/L This differs from the laboratory high sensitivity troponin assay. Any person depicted in such photographs is a model. Photos displayed are for illustrative purposes only. Cardiac troponin T increases rapidly 10 after. is a cardiospecific, highly sensitive marker for myocardial damage. Your use of this website and the information contained herein is subject to our Website Terms and Conditions and Privacy Policy. Patients who have been regularly exposed to animals or have received immunotherapy or diagnostic procedures using immunoglobulin or immunoglobulin fragments may produce antibodies, e.g. The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. This website is governed by applicable U.S. Incorporating i-STAT CHEM8+ early in the patient experience may help transform the delivery of care by. With i-STAT CHEM8 +, healthcare professionals can obtain chemistries, electrolytes, hematocrit and hemoglobin levels in approximately two minutes without leaving the patient's side. No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the company. Providing lab-quality results fast when every minute counts. Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics.©2023 Abbott. i-STAT 1 A handheld blood analyzer that delivers lab-quality, diagnostic results in minutes. 
GDPR Statement | Declaration for California Compliance Law. 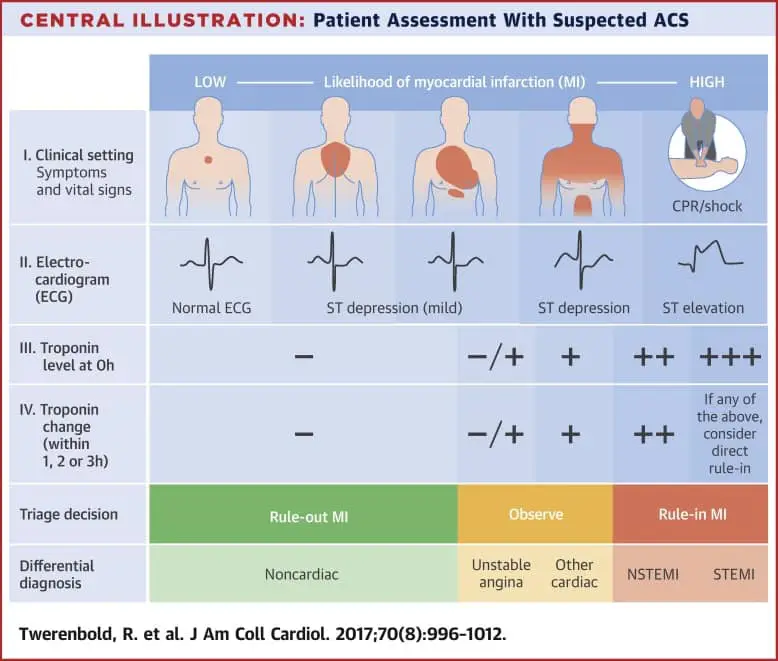
Your use of this website and the information contained herein is subject to our Website Terms and Conditions and Privacy Policy. The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. The i-STAT CG4+ cartridge with the i-STAT 1 System is intended for use in the in vitro quantification of pH, PO 2, PCO 2, and lactate in arterial or venous whole blood in point of care or clinical laboratory settings. No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the company. Background: Few studies have investigated the role of cardiac troponin point-of-care (POC) testing for predicting adverse outcomes in acute coronary syndrome (ACS) patients. Connects with your EMR system to enable seamless integration of results. Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. Delivers results in minutes no special sample preparation or user calibration needed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
